Peak Force Infrared (PFIR) Microscopy
Peak Force Infrared Microscopy enabling simultaneous chemical and mechanical imaging at ~6nm resolution.
What is Peak Force Infrared (PFIR) Microscopy?
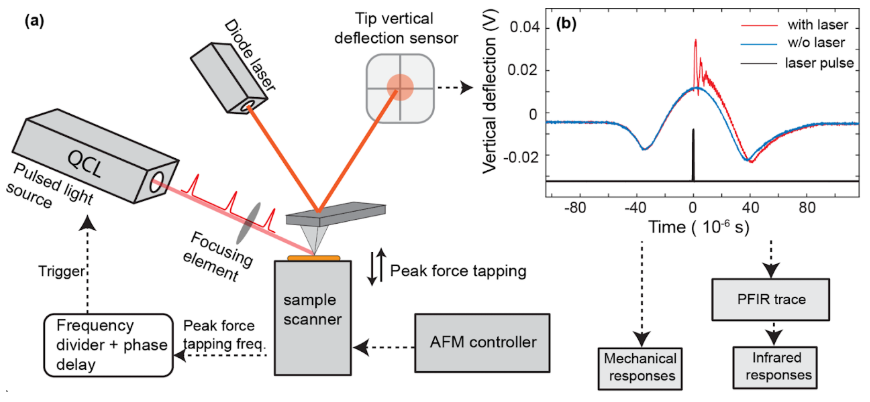
PFIR microscopy is action-based microscopy with mechanical detection of photothermal expansion by atomic force microscope (AFM) operated in the peak force tapping mode. It falls under the umbrella of the family of photothermal AFM-IR techniques. Our group at Lehigh University invented PFIR microscopy in 2016-2017.
The underlying mechanism for PFIR (and AFM-IR in general) is to leverage the high force sensitivity of an AFM cantilever to measure the mechanical effect of IR-specific photothermal expansion of the sample. The contact area of the AFM tip can be smaller than the tip radius in PFIR, thus the measurement is achieved from an area much smaller than the optical diffraction limit--a route for super-resolution microscopy.
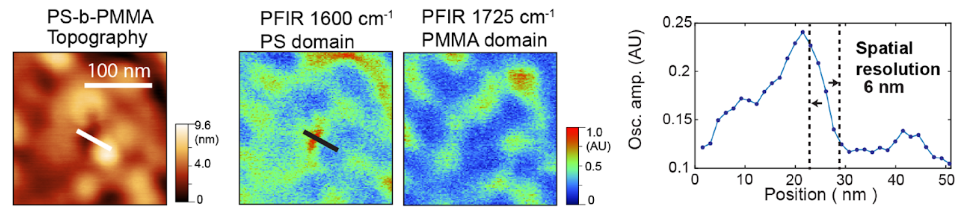
The unique trait of PFIR among AFM-IR techniques is the AFM tip is predictably in contact and detached from the sample surface under feedback loop control. The sample surface integrity is well preserved. As the cantilever resonances behave differently when in contact or detached from the sample, no residual photothermal signals are carried over from previous tip-sample interactions, thus ensuring no signal spillover between adjacent image pixels and achieving high spatial resolution as high as 6 nm.
The PFIR microscopy has another intrinsic advantage inherited from the peak force tapping mode. Under weak IR illumination, the mechanical properties of modulus and adhesion are simultaneously collected with the IR images, without separate scans. As the peak force tapping mode works well in the fluid, the PFIR microscopy also works in the fluid phase, including water.
The details of the PFIR microscopy are described in our recent Chemical Society Review article.
PFIR Achieves Label-free Chemical Imaging at 6 nm Spatial Resolution
The photothermal responses of the sample are typically extracted from the cantilever response using a data acquisition card, processed in real-time, and used as the PFIR signal. This approach achieves label-free chemical imaging at 6 nm spatial resolution, far surpassing the diffraction limit of conventional optical methods.
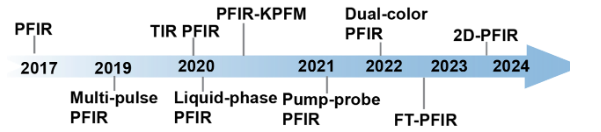
Improvements Since Invention
Since its invention, PFIR microscopy has been improved in several aspects:
- Multi-pulse PFIR for improvement of signal
- Total internal reflection and Liquid-phase PFIR microscopy
- Dual-color PFIR for multiple component imaging
- PFIR-KPFM for multimodal chemical, mechanical, and electrical imaging
If you are interested in measurement of your samples with PFIR, or building a PFIR microscope in your lab, please contact us. We are open to collaborations.
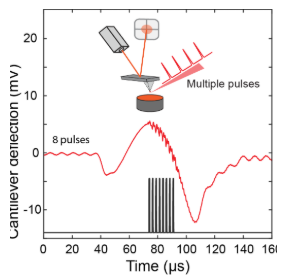

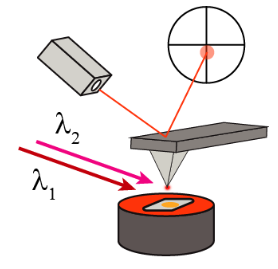
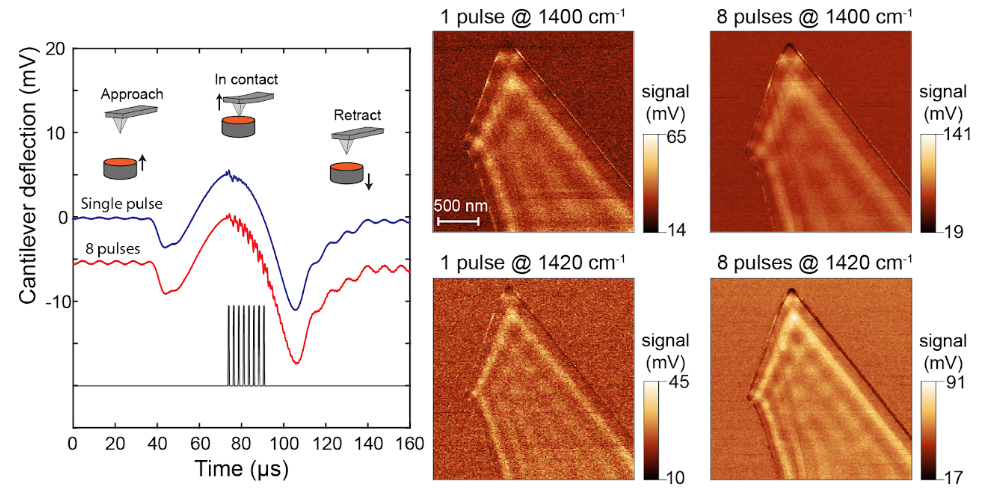
Multi-pulse PFIR Microscopy
One main limitation of the regular PFIR microscopy is its relatively weak signal. The peak force tapping cycle of an AFM is usually a few kHz (1kHz up to 8 kHz), which means that the tip and sample are in contact a few thousand times per second. At each tip-sample contact, a laser pulse is emitted to excite the photothermal expansion of the sample, and the cantilever responses are extracted to be used as a signal.
However, for samples with low photothermal responses or IR absorption, the PFIR signals are often weak.
The implementation of the multi-pulse PFIR is to illuminate multiple laser pulses per peak force tapping cycle. So more photothermal responses are generated per unit time. The resulting method improves the signal quality of PFIR microscopy.
The details of multi-pulse PFIR microscopy are described in our paper Advanced Optical Materials 1901084 (2019).
Liquid-Phase PFIR and Total Internal Reflection PFIR
One limitation of traditional infrared microscopy, aside from the restriction of spatial resolution from Abbe's diffraction limit, is its application in the aqueous phase. Water strongly absorbs mid-IR radiation, making the optical delivery difficult. Water absorption also strongly attenuates optical detection after light-matter interactions, reducing IR signals.
Infrared nanoscopy in aqueous has been a challenge.
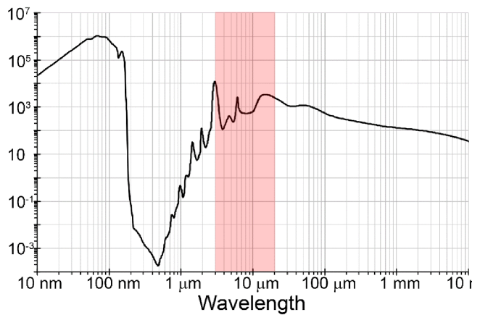
The action-based PFIR microscopy has several advantages in fluid operation:
- Spatial resolution is not limited by the diffraction limit
- Mechanical detection of photothermal effect bypasses the transmission attenuation after light-matter interactions
- The peak force tapping mode has been proven to operate well in the fluid phase
- Time-gated signal detection scheme is not drastically affected by the reduction of the cantilever quality factor
All these advantages make PFIR a promising method for aqueous phase nano-IR microscopy.
Dual-Color PFIR Microscopy
One possible extension of the PFIR-type photothermal detection mechanism is to incorporate multiple laser radiations with different beam characteristics (i.e., frequency, timing etc.) Such integration of multiple frequency excitations enables new capabilities for peak force photothermal microscopy.
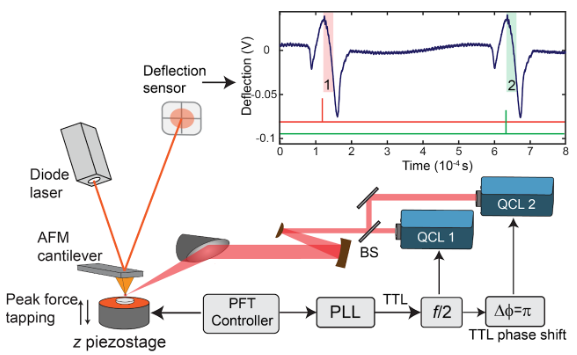
Dual-color PFIR microscopy is a variation of PFIR microscopy, in which two IR radiations of different frequencies are utilized to simultaneously deliver two PFIR images at different frequencies. The dual-color PFIR microscopy is good to image two chemical components of interest of non-sparse distributions, or with an open-loop AFM scanner, like the PeakForce Tapping enabled Multimode AFM of Bruker.
The dual-color PFIR microscopy is assembled with two separate mid infrared light sources (Quantum cascade laser QCL). The timing of the laser emission are synchronized to subsequent peak force tapping cycles. The photothermal responses are generated separately for each laser radiations and mechanically extracted with AFM cantilever oscillations.
Two signals from two IR excitations are simultaneously registered, together with the AFM topography and mechanical property channels.
The construction of dual-color PFIR microscope is described in our paper Analytical Chemistry, 94, 2, 1425-1431 (2022).
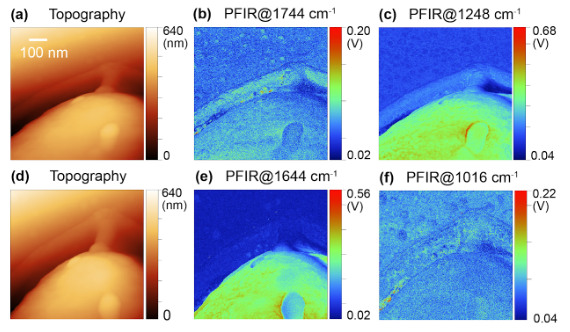
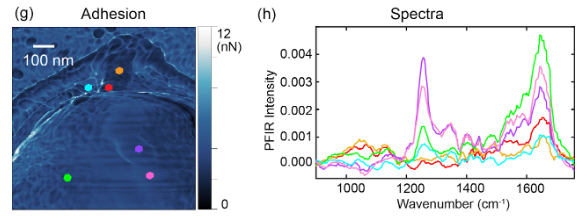
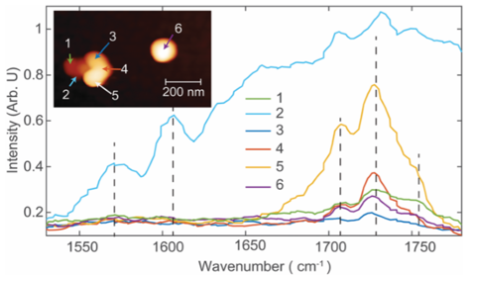
Applications of PFIR
We are actively seeking collaborations to apply PFIR microscopy. If you have a sample that requires nanoscale multimodal characterization, feel free to reach out to Dr. Xu.
Aerosols are tiny particles levitating in the air. Small aerosol particles (PM2.5, diameter < 2.5 microns) are difficult to be filtered out and can reach respiratory trait. Small aerosol particles also float in the air for many hours, even days. The main transmission route for COVID-19 pathogen is believed to be aerosols. The PFIR microscopy provides a route for both chemical and mechanical characterizations under ambient conditions.
- Urban aerosols: Spectra within urban collected aerosol particle. Oxidization signature is observed, suggesting secondary organic aerosol origin. Chemical Communications, 53, 7397 (2017)
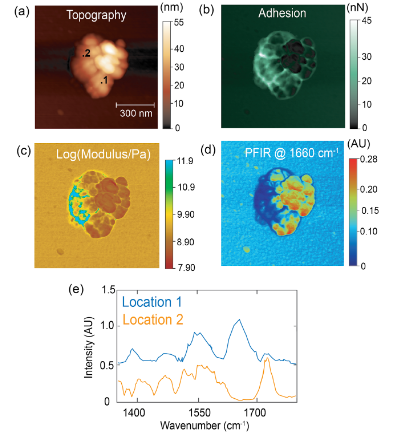
- Indoor aerosols: Multimodal imaging of an indoor aerosol particle, revealing both protein and carbonyl signatures. Analytical Chemistry, 93, 50, 16845-16852 (2021)
Key Features
- Simultaneous chemical and mechanical imaging
- ~6nm spatial resolution, far beyond diffraction limit
- Works in both air and liquid/aqueous phase
- Compatible with soft polymers and biological materials
- Multi-pulse mode for enhanced signal quality
- Multimodal capability with KPFM integration
Applications
- Aerosol particle analysis (PM2.5)
- Biological cell characterization
- Amyloid fibril protein studies
- Oil shale and kerogen mapping
- Phonon polaritons in h-BN
- Click chemistry in aqueous phase
Key Publications
View All Related PublicationsLock-in Amplifier Based Peak Force Infrared Microscopy
Andrea Dorsa, Qing Xie, Martin Wagner, and Xiaoji G. Xu
View Publication →What do different Modes of AFM-IR Mean for Measuring Soft Matter Surfaces?
Qing Xie and Xiaoji G. Xu
View Publication →